Pharmaceutics | Free Full-Text | Matrix Effects of the Hydroethanolic Extract of Calyces of Physalis peruviana L. on Rutin Pharmacokinetics in Wistar Rats Using Population Modeling

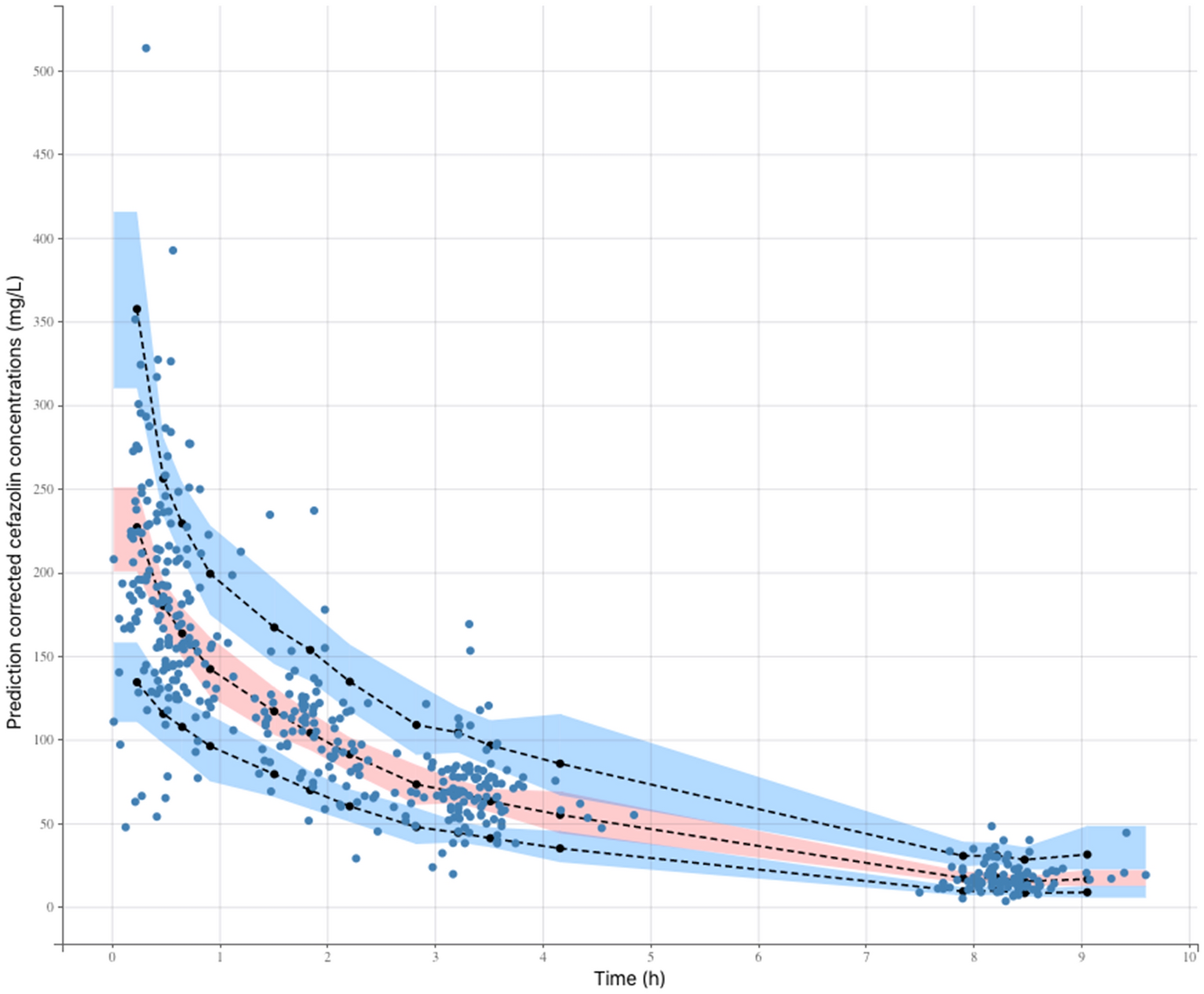
Simulation results using PK parameters obtained from PopPK analysis... | Download Scientific Diagram

Combining “Bottom‐up” and “Top‐down” Approaches to Assess the Impact of Food and Gastric pH on Pictilisib (GDC‐0941) Pharmacokinetics - Lu - 2017 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

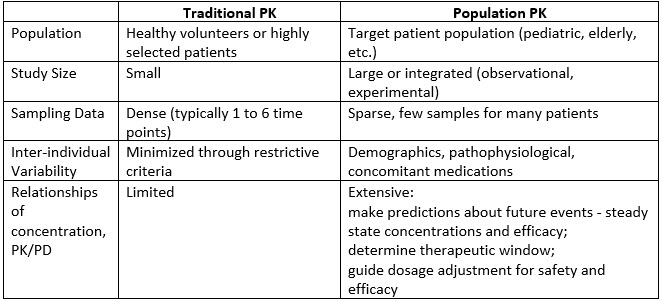
Clinical applications of population pharmacokinetic models of antibiotics: Challenges and perspectives - ScienceDirect

A Survey of Population Pharmacokinetic Reports Submitted to the USFDA: An Analysis of Common Issues in NDA and BLA from 2012 to 2021 | SpringerLink
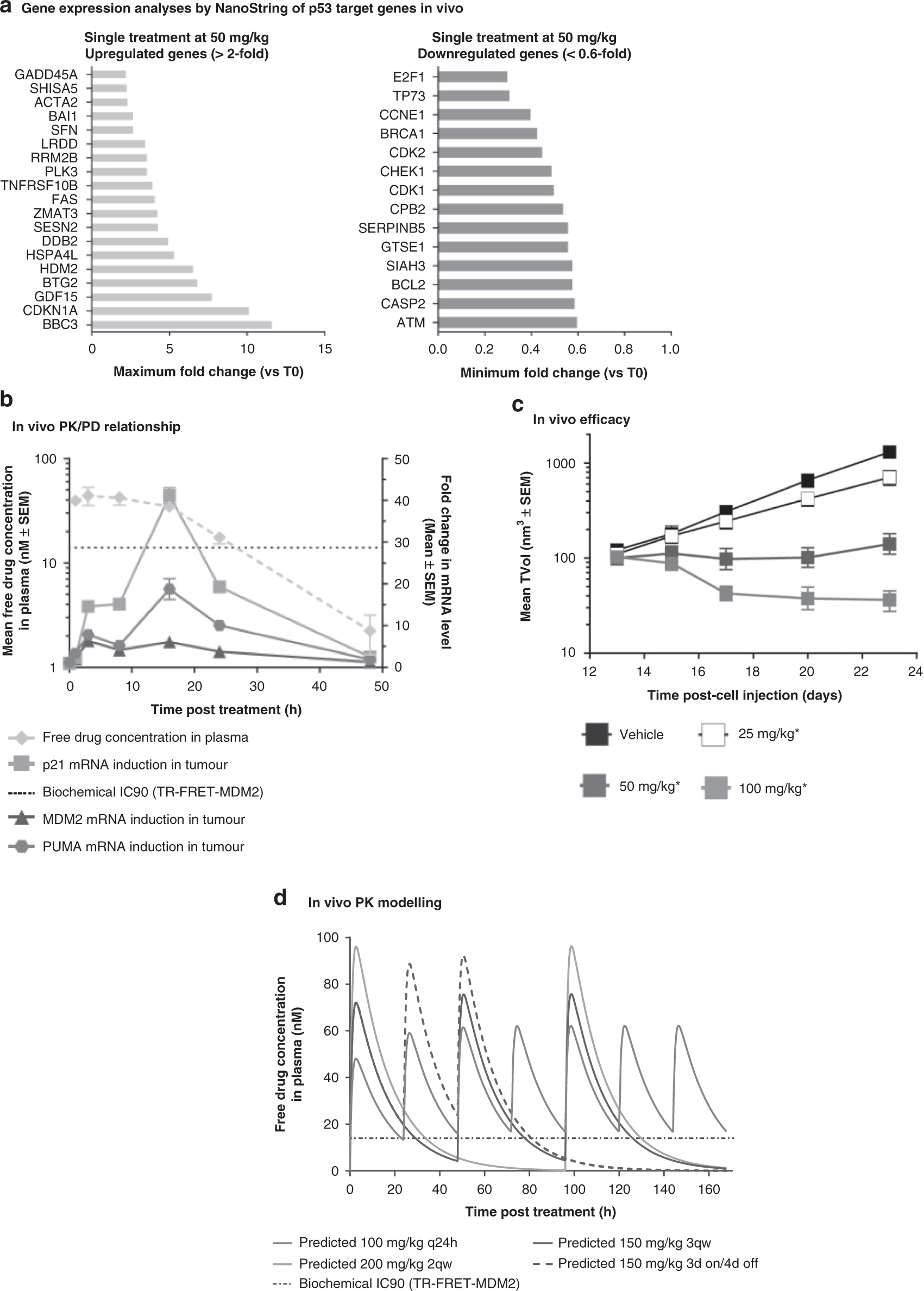
Pharmacokinetic–pharmacodynamic guided optimisation of dose and schedule of CGM097, an HDM2 inhibitor, in preclinical and clinical studies | British Journal of Cancer
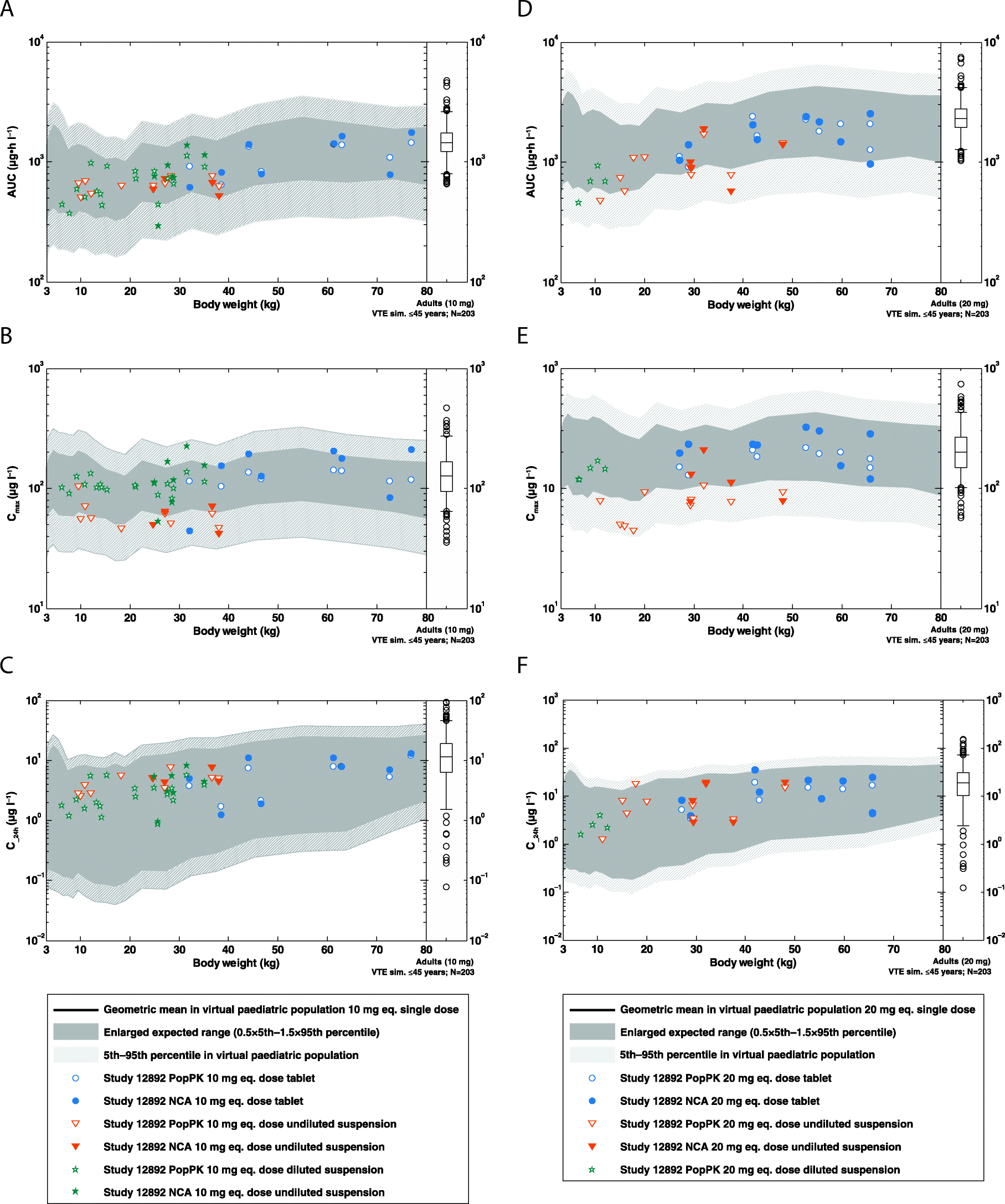
Pharmacokinetics of rivaroxaban in children using physiologically based and population pharmacokinetic modelling: an EINSTEIN-Jr phase I study | Thrombosis Journal | Full Text

Pharmacokinetics and population pharmacokinetics in pediatric oncology - Sassen - 2020 - Pediatric Blood & Cancer - Wiley Online Library
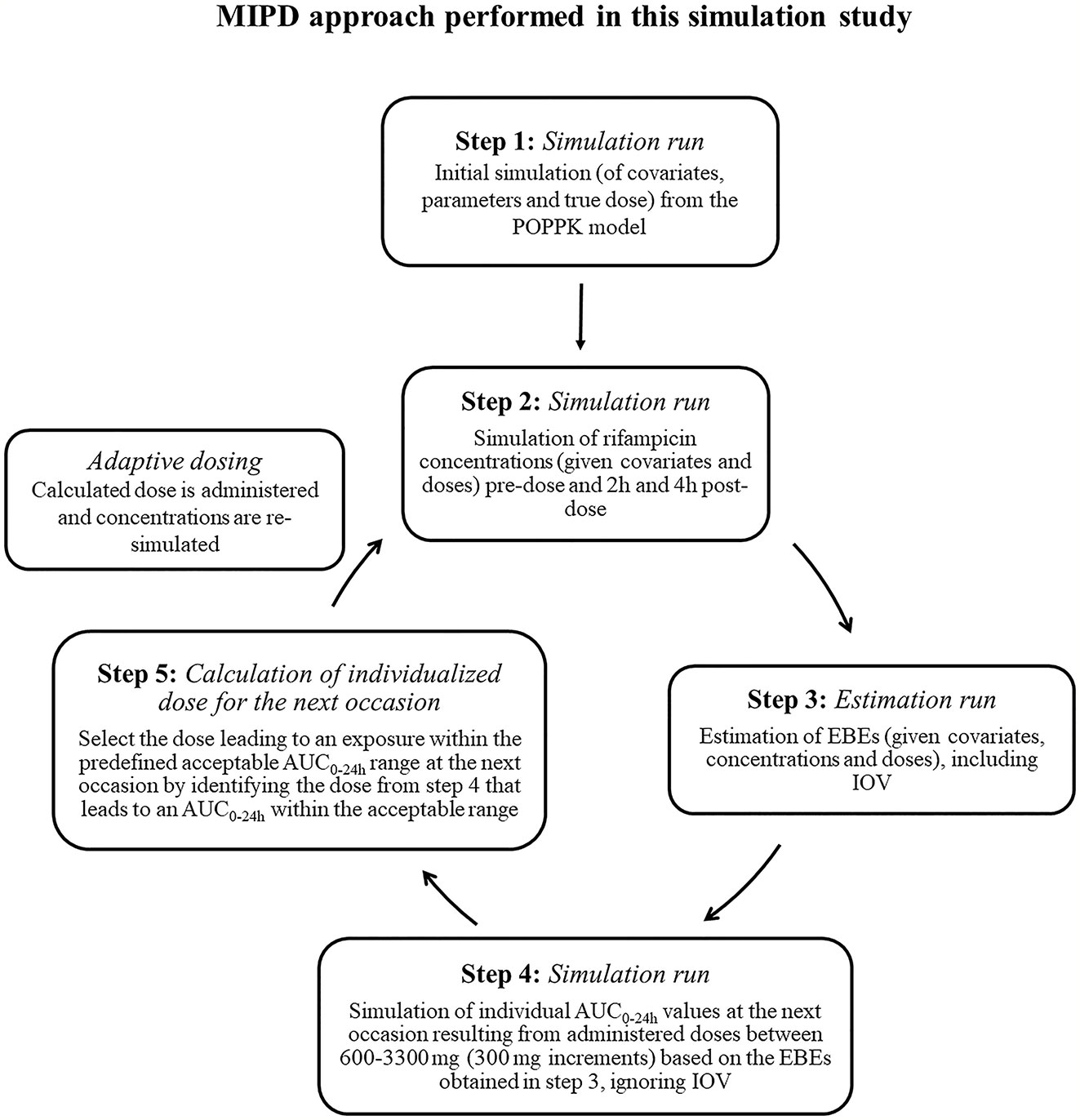
Frontiers | Individualized Dosing With High Inter-Occasion Variability Is Correctly Handled With Model-Informed Precision Dosing—Using Rifampicin as an Example

A Novel Integrated Pharmacokinetic-Pharmacodynamic Model to Evaluate Combination Therapy and Determine In Vivo Synergism | Journal of Pharmacology and Experimental Therapeutics

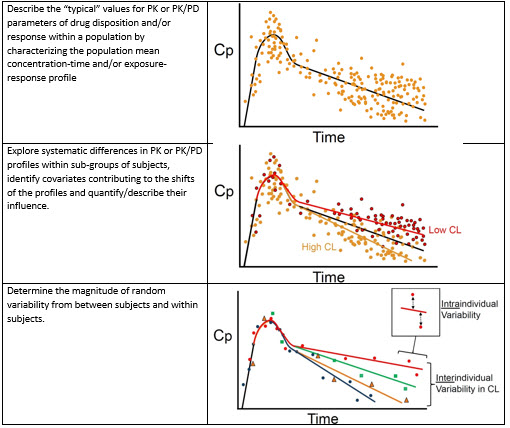
Establishing Best Practices and Guidance in Population Modeling: An Experience With an Internal Population Pharmacokinetic Analysis Guidance - Byon - 2013 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

Population Pharmacokinetic Variables: THETA, ETA, EPSILON; and NONMEM for analysis - Pharmacoengineering

Population pharmacokinetics and exposure-response analysis of sotrovimab in the early treatment of COVID-19 | medRxiv